-
Services
-
Practice Areas
- Banking and Finance
- Capital Markets
- Corporate M&A
- Dispute Resolution
- Employment and Labour
- EU and Competition Law
- Healthcare, Life Sciences & Pharmaceuticals
- Intellectual Property
- Projects and Energy
- Public Law
- Real Estate and Tourism
- Responsible Business
- Tax
- Technology, Media and Telecommunications
-
Sectors
- Agribusiness
- Banking and financial institutions
- Distribution and retail
- Energy and natural resources
- Government and public sector
- Healthcare, life sciences and pharmaceuticals
- Infrastructure
- Insurance and pension funds
- Manufacturing
- Mobility, transport and logistics
- Real estate and construction
- Social economy
- Sports
- Tourism and leisure
- Desks
- Buzz Legal
-
Practice Areas
-
People
-
Knowledge
-
Newsletter SubscriptionKeep up to date
Subscribe to PLMJ’s newsletters to receive the most up-to-date legal insights and our invitations to exclusive events.
-
-
About Us
-
Apply hereWe invest in talent
We are looking for people who aim to go further and face the future with confidence.
-
- ESG
-
Services
-
Practice Areas
- Banking and Finance
- Capital Markets
- Corporate M&A
- Dispute Resolution
- Employment and Labour
- EU and Competition Law
- Healthcare, Life Sciences & Pharmaceuticals
- Intellectual Property
- Projects and Energy
- Public Law
- Real Estate and Tourism
- Responsible Business
- Tax
- Technology, Media and Telecommunications
-
Sectors
- Agribusiness
- Banking and financial institutions
- Distribution and retail
- Energy and natural resources
- Government and public sector
- Healthcare, life sciences and pharmaceuticals
- Infrastructure
- Insurance and pension funds
- Manufacturing
- Mobility, transport and logistics
- Real estate and construction
- Social economy
- Sports
- Tourism and leisure
- Desks
- Buzz Legal
-
Practice Areas
-
People
-
Knowledge
-
Newsletter SubscriptionKeep up to date
Subscribe to PLMJ’s newsletters to receive the most up-to-date legal insights and our invitations to exclusive events.
-
-
About Us
-
Apply hereWe invest in talent
We are looking for people who aim to go further and face the future with confidence.
-
- ESG
Informative Note
New Rules on Cosmetic Products
26/03/2025The new law's main goal is to ensure the functioning of the internal market and to guarantee a high level of protection of human health.
The European legislator considered it necessary to establish a binding framework of rules directly applicable in all Member States due to the urgent need to harmonise rules.
On 19 March 2025, Decree-Law 23/2025 was published in the official gazette Diário da República. This new law incorporates Regulation (EC) 1223/2009 into Portuguese law and establishes the rules that cosmetic products placed on the market must comply with in order to ensure the functioning of the internal market and a high level of protection of human health (“Decree-Law 23/2025”).
There is an urgent need to harmonise rules in the Member States of the European Union in order to protect consumers and ensure the highest standards of human health protection. The European legislator therefore considered it necessary to establish a binding framework of rules directly applicable in all Member States.
To this end, Regulation (EC) 1223/2009 of 30 November 2009 lays down the standards to be met by cosmetic products placed on the market in order to ensure the functioning of the internal market and a high level of protection of human health.
Despite the application of Regulation (EC) 1223/2009, it is still possible for Member States to regulate the establishment of economic operators in the cosmetic products sector and the Regulation also gives Member States the power to regulate various matters.
Decree-Law 23/2025 therefore essentially provides that:
1. The provisions with which the establishment, operation and activity of economic operators in the cosmetic products sector must comply;
The manufacture, importation or first sale of cosmetic products in Portugal by an entity established in Portugal, other than the responsible person, is subject to registration with INFARMED, I. P. (the Portuguese National Authority of Medicines and Health Products)
It should also be noted that the loose supply of non-pre-packaged cosmetic products, cosmetic products packaged at the point of sale at the request of the purchaser or pre-packaged for immediate sale, may only be carried out by retailers and directly to the purchaser, excluding resale.
When importing cosmetic products from a third country to be placed on the market for the first time, or which have already been placed on the market but have undergone a change in formulation or any other modification that affects the conformity of the product, the importer of cosmetic products established in Portugal must submit to the customs authorities a declaration issued by INFARMED, I.P., certifying compliance with the regulatory framework established by Regulation (EC) 1223/2009 and Decree-Law 23/2025.
2. The mandatory information that must appear on the labelling of cosmetic products;
The labelling of cosmetic products must include the following information:
(a) The name or registered name and address of the responsible person;
(b) The nominal content at the time of packaging, given by weight or by volume;
(c) The date until which the cosmetic product, stored under appropriate conditions, will continue to fulfil its initial function;
(d) Particular precautions to be observed in use;
(e) The batch number of manufacture or the reference for identifying the cosmetic product;
(f) The function of the cosmetic product;
(g) A list of ingredients.
3. The language that should be used when writing the product information file;
Information on the nominal content of the product, the date until which the product continues to perform its functions, special precautions for use and the function of the product itself must be in Portuguese.
If the information has to be translated, the label attached to the product containing the translation must, in addition to being in indelible, legible and clearly visible characters, be affixed to the packaging container of the product, except in cases where, for practical reasons, it is not possible to include the necessary information on the label, e.g. because of the small size of the product. However, in such cases, the provision of the appropriate information is not waived, but may be provided by alternative means.
4. A system for reporting undesirable effects for health professionals, other professionals who use cosmetic products in their work and consumers themselves;
Similar to what already exists for medicines for human use in terms of obligations related to pharmacovigilance and the reporting of adverse reactions, Decree-Law 23/2025 provides for a similar system.
Consumers, health professionals and professionals established in Portugal who apply or use cosmetic products in the course of their professional activity can therefore notify INFARMED, I.P. of any serious or non-serious undesirable effects related to the use of cosmetic products.
Product managers and distributors have a real duty to report. Therefore, these operators must immediately inform INFARMED, I.P. of any serious undesirable effects they become aware of related to the use of cosmetic products for which they are responsible in Portugal.
5. An inspection system with its own measures to combat non-compliance with the obligations imposed by Decree-Law 23/2025 and Regulation (EC) 1223/2009;
INFARMED, I.P. is responsible for monitoring compliance with the provisions of Decree-Law 23/2025, as well as Regulation (EC) 1223/2009, and may even order:
(a) Corrective actions to make the cosmetic product compliant;
(b) The immediate recall, withdrawal or suspension of the sale of any cosmetic product or product wrongly classified as a cosmetic product;
(c) The issuance of guidelines on the conditions of use or of special accompanying measures, including warnings necessary to avoid risks to public health arising from the use of the cosmetic product;
(d) The imposition of special conditions or obligations on the entity that does not comply with the provisions of Decree-Law 23/2025, or other applicable legislation, and the setting of a deadline for rectifying the situation;
(e) Suspension of the exercise of the activity and/or closing of the establishment, including the respective manufacturing or storage facilities, in cases where this is justified in order to prevent or eliminate a situation of risk to public health.
6. A system of penalties that classifies non-compliance with the obligations imposed by Decree-Law 23/2025 and Regulation (EC) 1223/2009 as an administrative offence;
INFARMED, I.P. is also responsible for conducting, investigating and deciding on administrative offence proceedings arising from a market participant’s failure to comply with the obligations imposed by Decree-Law 23/2025 and Regulation (EC) 1223/2009.
In this context, it should also be noted that, compared to the previous regime established by Decree-Law 189/2008 of 24 September, there has been a significant reduction in the fines associated with very serious, serious and minorinfringements (other) breaches of these obligations.
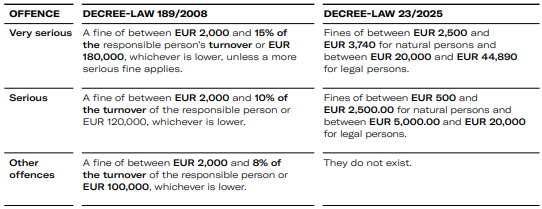
In any case, and taking into account the seriousness of the offence and the degree of fault of the perpetrator, INFARMED, I.P. may, if it so decides, impose a fine together with any other ancillary penalty provided for in the General Framework for Administrative Offences.
Decree-Law 23/2025 comes into force on 24 March 2025 and repeals Decree-Law 189/2008 of 24 September.