-
Services
-
Practice Areas
- Banking and Finance
- Capital Markets
- Corporate M&A
- Dispute Resolution
- Employment and Labour
- EU and Competition Law
- Healthcare, Life Sciences & Pharmaceuticals
- Intellectual Property
- Projects and Energy
- Public Law
- Real Estate and Tourism
- Responsible Business
- Tax
- Technology, Media and Telecommunications
-
Sectors
- Agribusiness
- Banking and financial institutions
- Distribution and retail
- Energy and natural resources
- Government and public sector
- Healthcare, life sciences and pharmaceuticals
- Infrastructure
- Insurance and pension funds
- Manufacturing
- Mobility, transport and logistics
- Real estate and construction
- Social economy
- Sports
- Tourism and leisure
- Desks
- Buzz Legal
-
Practice Areas
-
People
-
Knowledge
-
Newsletter SubscriptionKeep up to date
Subscribe to PLMJ’s newsletters to receive the most up-to-date legal insights and our invitations to exclusive events.
-
-
About Us
-
Apply hereWe invest in talent
We are looking for people who aim to go further and face the future with confidence.
-
- ESG
-
Services
-
Practice Areas
- Banking and Finance
- Capital Markets
- Corporate M&A
- Dispute Resolution
- Employment and Labour
- EU and Competition Law
- Healthcare, Life Sciences & Pharmaceuticals
- Intellectual Property
- Projects and Energy
- Public Law
- Real Estate and Tourism
- Responsible Business
- Tax
- Technology, Media and Telecommunications
-
Sectors
- Agribusiness
- Banking and financial institutions
- Distribution and retail
- Energy and natural resources
- Government and public sector
- Healthcare, life sciences and pharmaceuticals
- Infrastructure
- Insurance and pension funds
- Manufacturing
- Mobility, transport and logistics
- Real estate and construction
- Social economy
- Sports
- Tourism and leisure
- Desks
- Buzz Legal
-
Practice Areas
-
People
-
Knowledge
-
Newsletter SubscriptionKeep up to date
Subscribe to PLMJ’s newsletters to receive the most up-to-date legal insights and our invitations to exclusive events.
-
-
About Us
-
Apply hereWe invest in talent
We are looking for people who aim to go further and face the future with confidence.
-
- ESG
Informative Note
Use of medicines under off-label conditions
04/04/2025National Commission of Pharmacy and Therapeutics guidance note 19/March/2025
The National Commission of Pharmacy and Therapeutics (Comissão Nacional de Farmácia e Terapêutica – “CNFT”) approved Guidance Note 19/March/2025 on the use of medicines under off-label conditions on 14 March 2025.
Off-label use – the use of a medicine for an unapproved indication or in an unapproved age group, dosage or route of administration – is becoming increasingly important in clinical practice. This importance is justified by the complexity, length of time and financial cost associated with the process of technical and scientific evaluation of a medicine or a new therapeutic indication. The off-label use of medicines thus allows medicines to be used to treat pathologies for which the medicine does not have an approved therapeutic indication.
Off-label prescribing has become increasingly common and there are no specific regulations governing the practice. For this reason, the CNFT has considered it necessary to make a number of proposals, the main aim of which is to help standardise the actions of the Local Pharmacy and Therapeutics Commissions (Comissões de Farmácia e Terapêutica Locais – “Local CFTs”) when they receive requests for authorisation to use off-label medicines.
The CNFT therefore proposes:
A review of the conditions for the use of off-label medicines
The use of off-label medicines is restricted and depends on meeting all of the following conditions:
- There is no drug (active ingredient) with an approved indication for the patient's clinical situation, or the off-label use itself has a favourable risk-benefit ratio compared to the available alternatives;
- Off-label use is based on scientific evidence that therapeutic outcomes with a favourable risk-benefit ratio for the patient can be expected with the use of drugs in this regimen;
- A guarantee that the patient is properly monitored.
Classification of applications for off-label use
The CNFT also proposes that local CFTs classify applications for off-label use into three different groups, depending on the specific case and conditions of use:
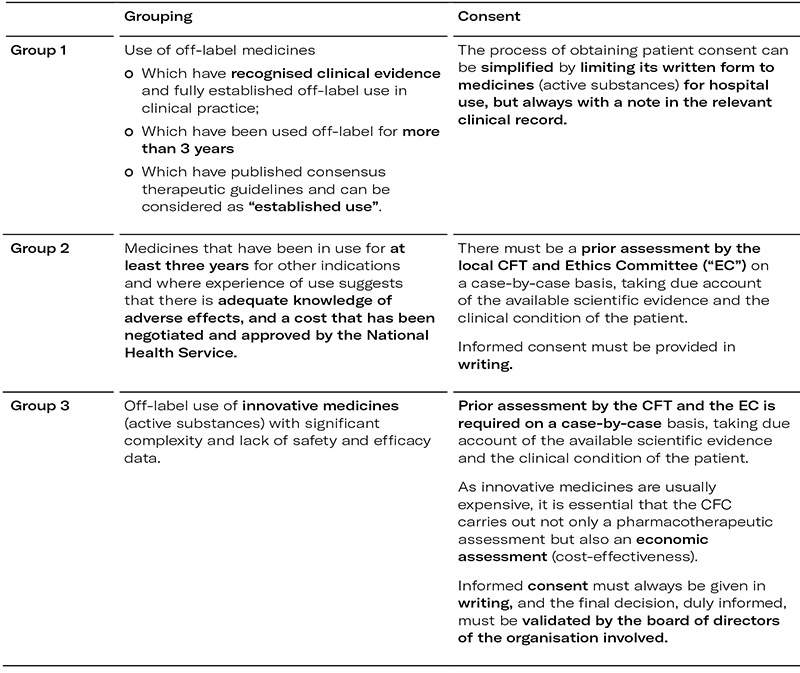
Conditions of the medicine supply chain for off-label prescriptions
In addition to the conditions on which the off-label use of medicines depends and the categorisation of each use, the CNFT also indicates the conditions that must be met by the medicine supply chain in order to issue the prescription in question:
- The prescription for use in a hospital or primary health care setting must indicate that it is for off-label use and this information must be recorded in the patient's clinical file;
- When authorised by the CFT, the scientific evidence or clinical consensus must be considered on a case-by-case basis in the specific context of the disease, the objectives to be achieved and the patient;
- Adequate patient monitoring must be ensured, with the clinical success and risk indicators to be recorded and the times for monitoring and informing the Local CFT being defined at the time of authorisation;
- The local decision-making process must be adapted to the degree of urgency, react in a timely manner and provide for exception mechanisms;
- When validating Group 1, the pharmacist must take into account the suitability of the off-label use of the medicine in accordance with locally approved therapeutic protocols. In the absence of a therapeutic protocol, applications must be submitted to the local CFT and EC for prior assessment;
- The pharmacist must have the necessary conditions to adequately inform the patient about the correct use by having access to the patient’s clinical information at the time of dispensing;
- The healthcare professional administering the medicine must be aware that it is an off-label use and of the need to monitor for unusual adverse reactions and the appropriate measures to control them.